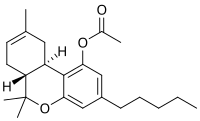
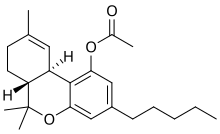
THC-O-acetate
 | |
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C23H32O3 |
| Molar mass | 356.506 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| | |
THC-O-acetate (THC acetate ester, O-acetyl-THC, THC-O, AcO-THC) is the acetate ester of THC. The term THC-O-acetate and its variations are commonly used for two types of the substance, dependent on which cannabinoid it is synthesized from. The difference between Δ8-THC and Δ9-THC is bond placement on the cyclohexene ring.[1]
Physical data, chemistry, and properties
THC acetate ester (THC-O or THCOA) can be synthesized from THC,[2][3] or from THCA. The acetylation of THC does not change the properties of the compound to the same extent as with other acetate esters, as the parent compound (THC) is already highly lipophilic, but potency is nonetheless increased to some extent. While the acetate ester of Δ9-THC is the best studied, the acetate esters of other isomers, especially Δ8-THC but also Δ10-THC are also known, as are other esters such as THC-O-propionate, THC-O-phosphate, THC hemisuccinate, THC hemiglutarate, THC morpholinylbutyrate, THC piperidinylpropionate,[4] THC naphthoyl ester (THC-NE),[5] and THC-VHS, as well as the hydrogenated derivative HHC-O-acetate and the ring-expanded Abeo-HHC acetate.
Pharmacology
It is a metabolic pro-drug, with its subjective effects being felt around 30 minutes after ingestion.[2][6]
Psychedelic claims
In a 2023 study, anecdotal claims surrounding THC-O-acetate's supposed ability to initiate psychedelic experiences were shown to not be significant. Answers using the Mystical Experience Questionnaire (MEQ) were under the threshold of a true experience, and those who had used classical psychedelics such as LSD or psilocybin consistently scored lower on the MEQ. When asked directly, 79% of the participants said it was either "not at all" or "a little" like a psychedelic experience.[7]
History
THC acetate ester was investigated as a possible non-lethal incapacitating agent as part of the Edgewood Arsenal experiments at some point between 1949 and 1975. It was noted to have about twice the capacity to produce ataxia (lack of voluntary coordination of muscle movements) as did THC when administered to dogs.[8]
Author D. Gold provided synthesis instructions for this compound (calling it "THC acetate") in his 1974 book Cannabis Alchemy: Art of Modern Hashmaking, in which it is described as follows;[2]
"The effect of the acetate is more spiritual and psychedelic than that of the ordinary product. The most unique property of this material is that there is a delay of about thirty minutes before its effects are felt."
The U.S. DEA first encountered THC-O-acetate as an apparent controlled substance analogue of THC in 1978. It was made in an analogous manner to how aspirin (acetylsalicylic acid) is made from willow bark (salicylic acid).[9] The incident was described by Donald A. Cooper of the DEA thus:
Given the world wide ready availability of marijuana, it is somewhat difficult to produce a viable argument for making [controlled substance analogs (CsA's)] of cannabinoids. However, ten years ago (1978) an attempt to produce CsA's from cannabis extracts was encountered in the Jacksonville, Florida area. In this case a concentrated extract of cannabis had been obtained by a soxhlet extraction. The extract had been acetylated with acetic anhydride, and in the final step, the excess acetic anhydride removed by distillation (reference is unretrievable due to its appearance in an underground periodical). The product contained neither quantities of nonderivatized cannabinoid nor any identifiable plant fragments. Since this single instance, no acetylated cannabinoid samples have been reported by a DEA laboratory. Therefore, this instance is assumed to represent an isolated occurrence and as such, will serve to terminate our discussion of cannabinoid CsA's.
A similar case reported in June 1995 in the United Kingdom.The description of that case appears to indicate the convicted manufacturer was using D. Gold's book Cannabis Alchemy as a guide.[10] THC acetate was also reported to have been found by New Zealand police in 1995, again made by acetylation of purified cannabis extracts with acetic anhydride.[11]
Following legal changes in the United States since around 2018, especially the legalisation of cannabis in an increasing number of states and the passage of the 2018 Farm Bill[12] which eased restrictions on the cultivation of industrial hemp, THC-O-acetate has become increasingly available as a recreational drug in the United States.[13]
Toxicity
In 2022, researchers at Portland State University screened for the presence of reacted ketene using N-benzylamine reported that Vitamin-E-acetate, CBD-di-acetate, CBN-O-acetate and THC-O-acetate may break down to release ketene gas when heated at 340 °C (644 °F). This study was unable to identify ketene itself and relied on reacting catalysts such as benzylamine which can also react with the acetic anhydride pre-cursor itself to form N-benzylacetamide as a false positive.[14]
In a follow-up study by the same authors (Kaelas Munger, Robert Jensen, Robert Strongin ), Acetic anhydride was confirmed to be the pre-cursor used and implicated a reaction scheme that would identify reacted N-Benzylacetamide from acetic anhydride separately from reacted N-Benzylacetamine from ketene. In this new study, a isotopically modified deuterium derivative CBN-O-Acetate-D3 was heated to 250 °C (482 °F) and formed reacted N-Benzylacetamide. The same study notes that ketene formation should not occur from vaping VEA, cannabinoid, or other acetates at real-world vaping temperature settings and that studies have shown that ketene is not expected to form from VEA or EtOAc, for example, at vaping temperatures under <700–850 °C. The same study notes that 3 other theoretical studies show that unrealistically high vaping temperatures, in excess of at least 700 °C, are required for any significant levels of ketene to form from VEA or other acetates.[15]
Legal status
United Kingdom
THC-O-acetate is a Class B drug in the United Kingdom.[16]
United States
THC-O-acetate is not listed under the Controlled Substances Act, meaning it is not scheduled substance.[17][18] A private letter from a DEA single representative which claimed that THC-O-Acetate met the statutory definition of ‘tetrahydrocannabinol on the basis that THC-O-Acetate is not known to occur in nature. This has been challenged in a federal court which resulted in rulings that THC-O-Acetate is legally derived from hemp. [17][19]
"(1) HEMP.—The term 'hemp' means the plant Cannabis sativa L. and any part of that plant, including the seeds thereof and all derivatives, extracts, cannabinoids, isomers, acids, salts, and salts of isomers, whether growing or not, with a Δ9-THC concentration of not more than 0.3 percent on a dry weight basis."[20]
In September 2024, a federal court once again ruled a binding decision that THC-O-Acetate is a legal substance if it's derived from hemp (Cannabis, cannabinoids, cannabinoids under 0.3% D9-THC).[21][22][23][24]
See also
References
- ↑ Sabaghi D (February 16, 2023). "Delta-8 And -9 THC-O Are Controlled Substances, DEA Says". Forbes.
- 1 2 3 Gold D (1974). Cannabis Alchemy: Art of Modern Hashmaking. Ronin Publishing (2010). ISBN 978-1-5795-1095-4.
- ↑ Starks M (1990). Marijuana Chemistry: Genetics, Processing, Potency. Ronin Publishing. ISBN 978-0-9141-7139-3.
- ↑ Banerjee A, Hayward JJ, Trant JF (May 2023). ""Breaking bud": the effect of direct chemical modifications of phytocannabinoids on their bioavailability, physiological effects, and therapeutic potential" (PDF). Organic & Biomolecular Chemistry. 21 (18): 3715–3732. doi:10.1039/d3ob00068k. PMID 36825573.
- ↑ Elkarim NS. Stable formulations of dronabinol. Patent US20210251947
- ↑ Starks M (1977). Marijuana Potency. And/Or Press. p. 174. ISBN 978-0-915904-27-3. Retrieved 3 April 2023.
- ↑ Kruger DJ, Bone CC, Meacham MC, Klein C, Kruger JS (June 2023). "THC-O-Acetate: Scarce Evidence for a Psychedelic Cannabinoid". Journal of Psychoactive Drugs. 56 (4): 563–567. doi:10.1080/02791072.2023.2230573. PMID 37381980. S2CID 259285965.
- ↑ Committee on Toxicology, National Research Council (1984). Possible Long-Term Health Effects of Short-Term Exposure To Chemical Agents, Volume 2: Cholinesterase Reactivators, Psychochemicals and Irritants and Vesicants. The National Academies Press. p. 79. ISBN 978-0-309-07772-9.
- ↑ Cooper DA. "Future Synthetic Drugs of Abuse". Drug Enforcement Administration. McLean, Virginia.
- ↑ Brown DT (2003). Cannabis: The Genus Cannabis. Hardwood Academic Publishers. p. 82. ISBN 90-5702-291-5.
- ↑ Valentine MD (1995). "Δ9-THC acetate from acetylation of cannabis oil". Science and Justice. 36 (3): 195–197. doi:10.1016/S1355-0306(96)72595-9.
- ↑ "President Trump Signed the Farm Bill into Law One Year Ago Today, USDA Highlights Implementation Accomplishments to Date" (Press release). Washington D.C.: USDA Press. USDA. 2019-12-20. Retrieved 2023-06-22.
- ↑ Holt AK, Poklis JL, Peace MR (October 2022). "∆8-THC, THC-O Acetates and CBD-di-O Acetate: Emerging Synthetic Cannabinoids Found in Commercially Sold Plant Material and Gummy Edibles". Journal of Analytical Toxicology. 46 (8): 940–948. doi:10.1093/jat/bkac036. PMC 9564187. PMID 35674405.
- ↑ Munger KR, Jensen RP, Strongin RM (July 2022). "Vaping Cannabinoid Acetates Leads to Ketene Formation". Chemical Research in Toxicology. 35 (7): 1202–1205. doi:10.1021/acs.chemrestox.2c00170. PMID 35801872.
- ↑ Munger KR, Anreise KM, Jensen RP, Peyton DH, Strongin RM (2024). "Mechanistic Rationale for Ketene Formation during Dabbing and Vaping". Jacs Au. 4 (6): 2403–2410. doi:10.1021/jacsau.4c00436.
- ↑ "The Misuse of Drugs Act 1971 (Amendment) Order 2008". Office of Public Sector Information. Retrieved 15 June 2009.
- 1 2 Sabaghi D. "Delta-8 And -9 THC-O Are Controlled Substances, DEA Says". Forbes. Retrieved 2023-07-14.
- ↑ "Response Letter: Control status under the Controlled Substances Act (CSA) of THC acetate ester (THCO)" (PDF). U. S. Department of Justice, Drug Enforcement Administration. 13 February 2023.
- ↑ "Controlled Substances, Alphabetical Order" (PDF). Orange Book. Diversion Control Division, Drug Enforcement Agency, U.S. Department of Justice.
- ↑ "7 U.S. Code § 1639o - Definitions".
- ↑ "Federal circuit court rules 'all products' made from federally compliant hemp are legal". 5 September 2024.
- ↑ "Federal Court Rules That THC-O is Legal Under 2018 Farm Bill in Surprise Win for Synthetic Cannabinoid Sellers". 6 September 2024.
- ↑ "THC-O Qualifies as Legal Hemp Under Federal Law, Appeals Court Says, Rejecting DEA's Restrictive Stance". 5 September 2024.
- ↑ "4th Circuit Challenges DEA's Findings on the Legality of Certain Hemp Derived Cannabinoids".