Triampyzine
 | |
| Clinical data | |
|---|---|
| Other names | (Dimethylamino)trimethylpyrazine; W-3976B; W3976-B; W-3976-B; W3976B; 3,5,6-Trimethylampyzine |
| Identifiers | |
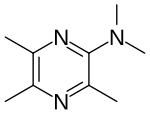
IUPAC name
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C9H15N3 |
| Molar mass | 165.240 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
Triampyzine (INN), also known as triampyzine sulfate (USAN; developmental code name W-3976B) in the case of the sulfate salt, as (dimethylamino)trimethylpyrazine, or as 3,5,6-trimethylampyzine, is a drug described as an anticholinergic and antisecretory agent which was never marketed.[1][2][3][4][5] It was first described in the literature by 1966.[1][6] The drug is the 3,5,6-trimethylated derivative of ampyzine (W-3580B), which is also a drug and is, conversely, described as a "central stimulant".[1][5][2]
References
- 1 2 3 Elks, J. (2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer US. p. 436. ISBN 978-1-4757-2085-3. Retrieved 21 October 2024.
- 1 2 Milne, G.W.A. (2018). Drugs: Synonyms and Properties. Routledge Revivals. Taylor & Francis. pp. 213, 547. ISBN 978-1-351-78990-5. Retrieved 21 October 2024.
- ↑ "TRIAMPYZINE". Inxight Drugs. Retrieved 21 October 2024.
- ↑ Negwer, M. (1994). Organic-chemical Drugs and Their Synonyms: (an International Survey). Akademie Verlag. p. 355. ISBN 978-3-05-500156-7. Retrieved 21 October 2024.
- 1 2 Lednicer, D. (2009). Strategies for Organic Drug Synthesis and Design. Wiley. p. 366. ISBN 978-0-470-39959-0. Retrieved 21 October 2024.
- ↑ Marler, E.E.J. (1967). Pharmacological and Chemical Synonyms: A Collection of Names of Drugs and Other Compounds Drawn from the Medical Literature of the World. Excerpta Medica. p. 322. Retrieved 21 October 2024.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.