Levosulpiride
 | |
| Clinical data | |
|---|---|
| Trade names | Dislep, Sulpepta, others |
| Other names | L-Sulpiride; S-(–)-Sulpiride; RV-12309 |
| Routes of administration | Oral |
| ATC code | |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
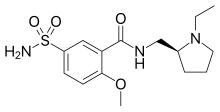
| Formula | C15H23N3O4S |
| Molar mass | 341.43 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
Levosulpiride, sold under the brand names Dislep and Sulpepta among others, is a dopamine antagonist medication which is used in the treatment of psychotic disorders like schizophrenia, major depressive disorder, nausea and vomiting, and gastroparesis.[1][2][3][4] It is taken by mouth.
It is a selective antagonist of the dopamine D2 receptor and an agonist of the serotonin 5-HT4 receptor.[4][5] Chemically, it is a benzamide and the (S)-(−)-enantiomer of sulpiride.[4]
Levosulpiride is marketed widely throughout the world, including in Europe, South Korea, Latin America, India, and Pakistan.[2] It is not available in the United States or the United Kingdom.[2]
Medical uses
Levosulpiride is used in the treatment of:[3][1]
- Psychosis
- Negative symptoms of schizophrenia
- Major depressive disorder
- Anxiety disorders
- Dysthymia
- Vertigo
- Nausea and vomiting
- Gastroparesis
- Dyspepsia
- Irritable bowel syndrome
- Premature ejaculation[6][7]
Levosulpiride is not currently licensed for treatment of premature ejaculation in the United Kingdom or other European countries.[8]
Side effects
Side effects of levosulpiride include amenorrhea, gynecomastia, galactorrhea, changes in libido, and neuroleptic malignant syndrome.[9] In the United States, as of 2013 only one case of adverse reaction to levosulpiride had been recorded on the FDA Adverse Event Reporting System Database.[8] A case of rapid-onset resistant dystonia caused by low-dose levosulpiride was reported in India.[10]
Pharmacology
Pharmacodynamics
Levosulpiride is a selective dopamine D2 receptor antagonist.[4] The drug has also been found to act as a moderate agonist of the serotonin 5-HT4 receptor.[5] It is said to have antipsychotic, antidepressant, antiemetic, and gastroprokinetic effects.[4]
Chemistry
Levosulpiride is a substituted benzamide derivative.[4] It is the levorotatory enantiomer of sulpiride.[4] Other benzamide derivatives include amisulpride, metoclopramide, tiapride, sultopride, and veralipride, among others.
References
- 1 2 "Levosulpiride". AdisInsight. 24 October 2021. Retrieved 22 October 2024.
- 1 2 3 "Levosulpiride (International database)". Drugs.com. 6 October 2024. Retrieved 22 October 2024.
- 1 2 Mucci A, Nolfe G, Maj M (February 1995). "Levosulpiride: a review of its clinical use in psychiatry". Pharmacol Res. 31 (2): 95–101. doi:10.1016/1043-6618(95)80053-0. PMID 7596960.
- 1 2 3 4 5 6 7 Rossi F, Forgione A (February 1995). "Pharmacotoxicological aspects of levosulpiride". Pharmacol Res. 31 (2): 81–94. doi:10.1016/1043-6618(95)80052-2. PMID 7596959.
- 1 2 Tonini M, De Giorgio R, Spelta V, Bassotti G, Di Nucci A, Anselmi L, Balestra B, De Ponti F (April 2003). "5-HT4 receptors contribute to the motor stimulating effect of levosulpiride in the guinea-pig gastrointestinal tract". Dig Liver Dis. 35 (4): 244–250. doi:10.1016/s1590-8658(03)00061-6. PMID 12801035.
- ↑ Arshad A, Irfan M, Inam M, Hussain NH, Ismail SB (2022). "Levosulpiride for Premature Ejaculation: A Systematic Review and Meta-Analysis". Am J Mens Health. 16 (5): 15579883221124832. doi:10.1177/15579883221124832. PMC 9515538. PMID 36154321.
- ↑ Greco E, Polonio-Balbi P, Speranza JC (August 2002). "Levosulpiride: a new solution for premature ejaculation?". Int J Impot Res. 14 (4): 308–309. doi:10.1038/sj.ijir.3900901. PMID 12152121.
- 1 2 Poluzzi E, Raschi E, Koci A, Moretti U, Spina E, Behr ER, et al. (June 2013). "Antipsychotics and torsadogenic risk: signals emerging from the US FDA Adverse Event Reporting System database". Drug Safety. 36 (6): 467–79. doi:10.1007/s40264-013-0032-z. PMC 3664739. PMID 23553446.
- ↑ "Levosulpiride drug information". DrugsUpdate India.
- ↑ Naskar S, Nath K (January 2007). "Rapid onset resistant dystonia with low dose of Levosulpiride". British Journal of Psychiatry. 190 (1): 81. doi:10.1192/bjp.190.1.81a.